Importance of Optimum Soil pH
- Published in
- Category: Specialist Articles
- Hits: 6228
- Print , Email
Why is soil pH important?
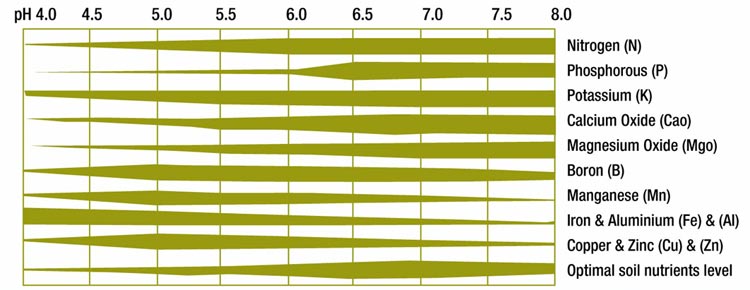
Soil pH plays an important role in determining the availability of nutrients to a plant.
At a pH < 6.0, the phosphorous present in the soil is 'fixed' and not available to the plant.
At a pH < 4.5, heavy metal and aluminium toxicity can occur, resulting in stunted growth.
At a pH > 7.5, the availability of micro nutrients and essential minerals such as Zinc, Copper,
Boron and Molybdenum are severely limited.
|
|
| Soil displaying optimum pH levels sustain healthy crops and produce rich yield. |
Optimum soil pH
Apart from plants which thrive in highly acidic soil, the optimum soil pH for the majority of plants is between 6.0 and 6.5. Soil featuring these pH levels are particularly conducive to nutrient uptake; fertilizers applied to soil displaying these pH levels demonstrate the best improved results.
In tropical conditions, most of the soil present display acidic properties. As such, monitoring and ensuring that the soil pH is kept at the optimum level is essential for healthy growth, good yield and promotes land sustainability.
Optimum soil pH chart
 |



